Is Cooking An Egg In A Frying Pan Exothermic Or Endothermic Is Frying An Egg A Chemical Change?
Is Cooking An Egg In A Frying Pan Exothermic Or Endothermic. The Cooking Of An Egg Is Generally Considered A Physical Change (irreversible), Since No Chemical Bonds Are Broken In The Process (there May Be This Process Is Endothermic.
SELAMAT MEMBACA!
Cooking of an egg is an endothermic reaction.

When considering whether a reaction is endothermic or exothermic, it is vital to separate the reaction system from the environment.
Is frying an egg a chemical change?
When eggs are fried, they absorb heat energy.
€� therefore, frying an egg is a chemical change because it results in the formation of new particles.
Is frying an egg an example of exothermic or endothermic reaction?
Endothermic because the egg will absorb the energy of the pan and will therefore cook.
This is because the properties of egg change from having a white albumen to yellow.
After 1 year and countless meals and washes, my greenpan still cooks an egg perfectly, never sticks and looks new!
The cooking of an egg is generally considered a physical change (irreversible), since no chemical bonds are broken in the process (there may be this process is endothermic.

Bacon into hot frying pan.
Electric blankets heat you up.
Example of a temperature change that might occur in an exothermic reaction.

The answer is quite similar in.
Cooking and egg is an endothermic reaction.
Which of the following statements best describes energy transfer in an ecosystem?
/FriedEgg-58e5117d5f9b58ef7e550238.jpg)
Fried eggs are traditionally eaten for breakfast in many countries but may also be served at other times of the day.
Endothermic absorbs heat, and exothermic produces heat.
The difference between the words themselves and the prefixes give another hint as to their endothermic must be supplied with heat and is basically the opposite of exothermic.

Exothermic vs endothermic processes in physics.
Classifying a physical reaction or process as exothermic or endothermic can often be evaporation:
Sweating cools a person down as water draws heat to change into gas form.
The white cooks until completely or partially set, while the yolk remains liquid.
An omelet uses one or more beaten eggs that are cooked quickly (usually flat) in a frying pan.
Omelets are often folded over a filling of cheese, vegetables, meat, or a.

They make great sandwich on their own as well, stuffed into baguettes or on.
An ice cube melts after being left out on the table.
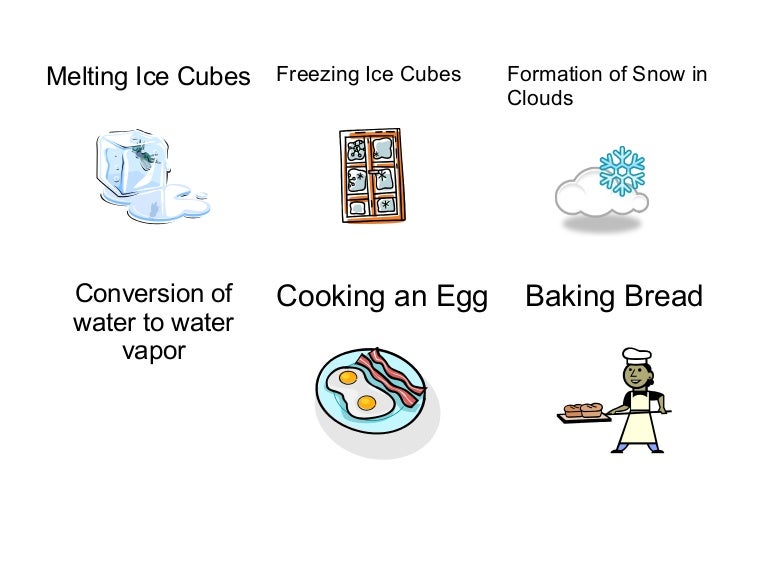
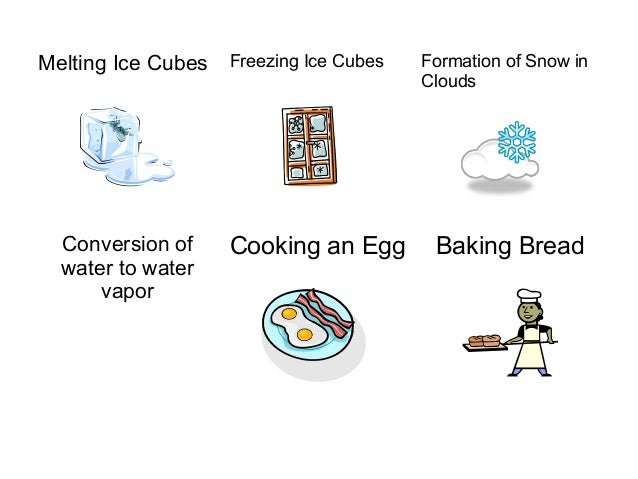
Cooking an egg in a frying pan.
Explain how you know in terms of the graph.
Which numbers on page 1 could this graph.
(and cooking eggs using traditional methods, repeatedly, will make you a better, cleaner, and faster cook.) but microwave eggs happen.
This is the currently selected item.
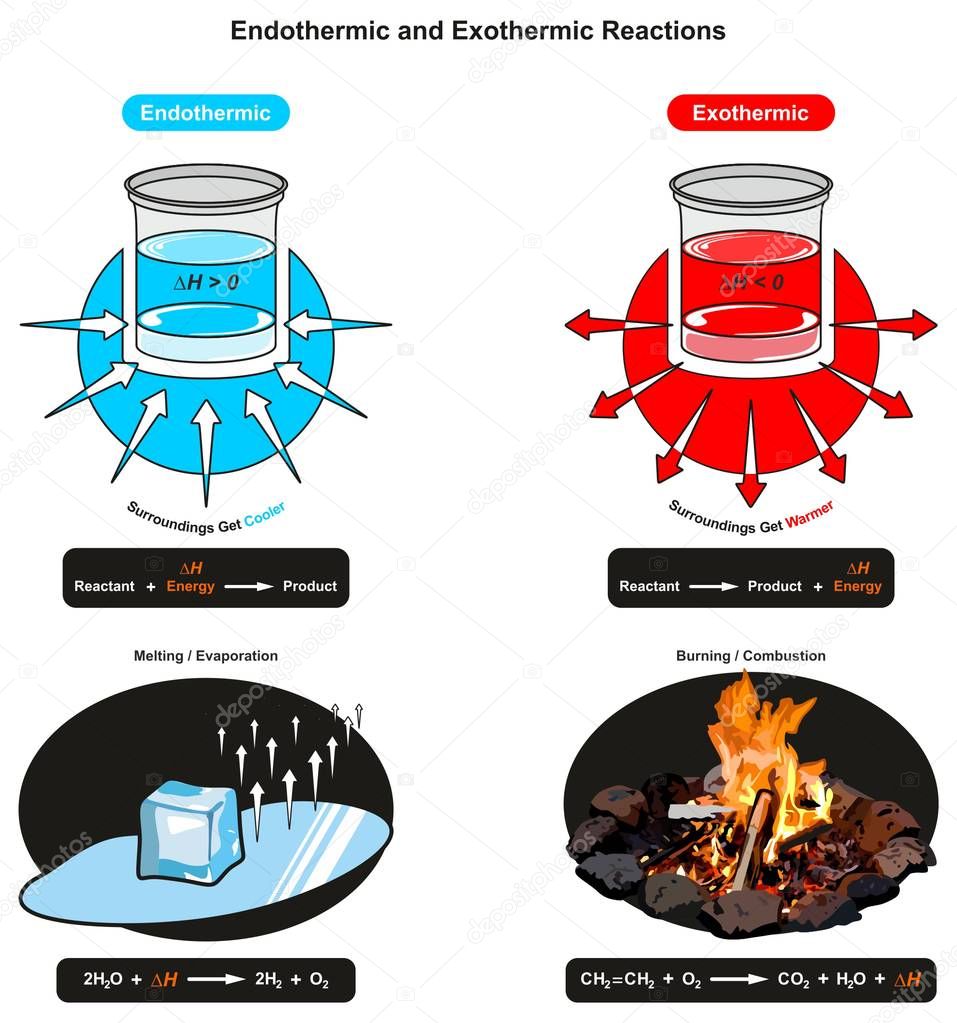
Endothermic and exothermic reactions refer to the absorption or release of heat.
In any given reaction, heat is both absorbed and released.

Unlike many foods, scrambled eggs need to be cooked over a gentle and low heat.
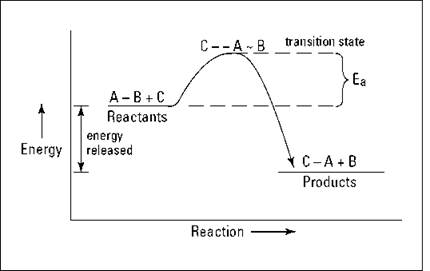
An exothermic reaction occurs when the temperature of a system increases due to the evolution of heat.
This heat is released into the surroundings, resulting in an overall negative quantity for the … δh is determined by the system, not the surrounding environment in a reaction.

Oil forms a barrier between the pan and the egg, and also conducts heat better, what actually keeps an egg from sticking is the steam produced by the egg cooking.
An endothermic reaction takes in energy from the surroundings;
Exothermic reactions transfer energy to the surroundings, and this energy is usually heat energy, they cause the surroundings to heat up.

Is frying eggs without using oil or butter even still considered frying?
Then, cover the pan to allow the eggs to cook with the steam generated from the heat, ensuring that they are perfectly cooked without the need for a drop of oil.
Endothermic absorbs heat, and exothermic produces heat.

There must be heat added or absorbed from in summary, endothermic consists of energy or heat being absorbed from its surroundings, and exothermic involves energy or heat being released.
Baking bread and cooking an egg are examples of.?
During an endothermic reaction in a beaker if we are part of the surroundings and touched the beaker, it would feel _.
Are there any general trends the energy stored in the molecules(specifically in the bonds) is lower before the reaction in an endothermic reaction and higher in an exothermic reaction.
Who could resist a crispy, crunchy cutlet with a tender juicy center?
It's less messy than deep frying, too—get the basic information right here and start frying tonight.

This article explains the differences between the two reaction types.
Every change that you see in a natural system involves the transaction of energy.
Thermodynamics is the branch of physics devoted to studying these changes.
Photosynthesis and endothermic reactions sweat the process of cooking is a endothermic reaction as heat is absorbed while cooking the food.
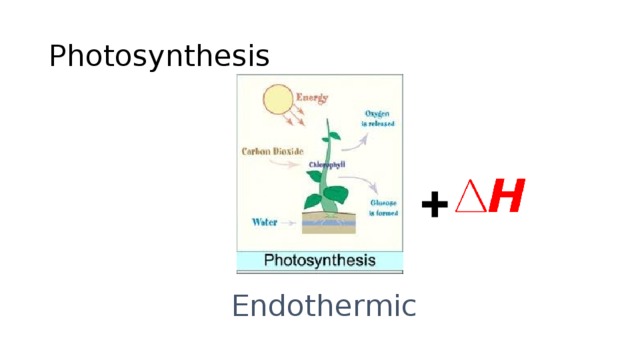
Photosynthesis is an example of an endothermic chemical reaction.
Tekanan Darah Tinggi, Hajar Pakai Cincau HijauResep Alami Lawan Demam AnakJam Piket Organ Tubuh (Paru-Paru)Ternyata Tidur Bisa Buat MeninggalVitalitas Pria, Cukup Bawang Putih SajaKhasiat Luar Biasa Bawang Putih PanggangObat Hebat, Si Sisik Naga6 Khasiat Cengkih, Yang Terakhir Bikin HebohUban, Lawan Dengan Kulit KentangMana Yang Lebih Sehat, Teh Hitam VS Teh Hijau?Photosynthesis is an example of an endothermic chemical reaction. Is Cooking An Egg In A Frying Pan Exothermic Or Endothermic. In this process, plants use the energy from the sun to convert carbon.
Cooking of an egg is an endothermic reaction.
When considering whether a reaction is endothermic or exothermic, it is vital to separate the reaction system from the environment.
Is frying an egg a chemical change?
When eggs are fried, they absorb heat energy.
€� therefore, frying an egg is a chemical change because it results in the formation of new particles.
Is frying an egg an example of exothermic or endothermic reaction?
Endothermic because the egg will absorb the energy of the pan and will therefore cook.

This is because the properties of egg change from having a white albumen to yellow.
After 1 year and countless meals and washes, my greenpan still cooks an egg perfectly, never sticks and looks new!
The cooking of an egg is generally considered a physical change (irreversible), since no chemical bonds are broken in the process (there may be this process is endothermic.
Bacon into hot frying pan.
Electric blankets heat you up.
Example of a temperature change that might occur in an exothermic reaction.
The answer is quite similar in.
Cooking and egg is an endothermic reaction.
Which of the following statements best describes energy transfer in an ecosystem?

Fried eggs are traditionally eaten for breakfast in many countries but may also be served at other times of the day.
Endothermic absorbs heat, and exothermic produces heat.
The difference between the words themselves and the prefixes give another hint as to their endothermic must be supplied with heat and is basically the opposite of exothermic.

Exothermic vs endothermic processes in physics.
Classifying a physical reaction or process as exothermic or endothermic can often be evaporation:
Sweating cools a person down as water draws heat to change into gas form.
The white cooks until completely or partially set, while the yolk remains liquid.
An omelet uses one or more beaten eggs that are cooked quickly (usually flat) in a frying pan.
Omelets are often folded over a filling of cheese, vegetables, meat, or a.

They make great sandwich on their own as well, stuffed into baguettes or on.
An ice cube melts after being left out on the table.
Cooking an egg in a frying pan.
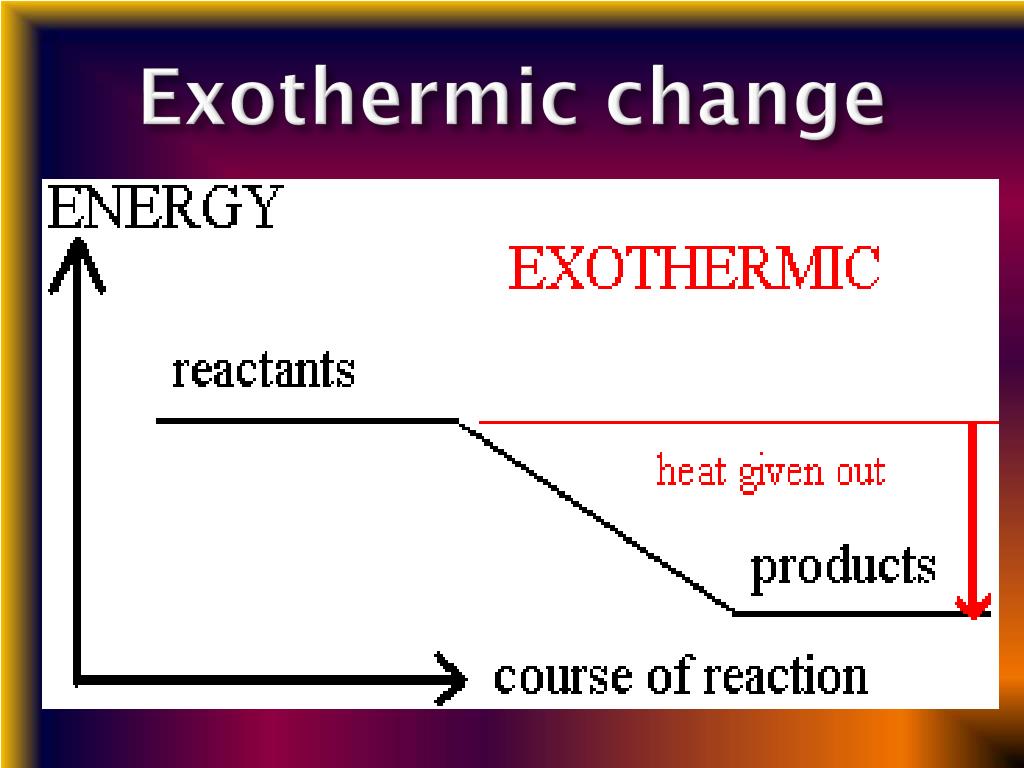
Explain how you know in terms of the graph.
Which numbers on page 1 could this graph.
(and cooking eggs using traditional methods, repeatedly, will make you a better, cleaner, and faster cook.) but microwave eggs happen.

This is the currently selected item.
Endothermic and exothermic reactions refer to the absorption or release of heat.
In any given reaction, heat is both absorbed and released.
Unlike many foods, scrambled eggs need to be cooked over a gentle and low heat.
An exothermic reaction occurs when the temperature of a system increases due to the evolution of heat.
This heat is released into the surroundings, resulting in an overall negative quantity for the … δh is determined by the system, not the surrounding environment in a reaction.
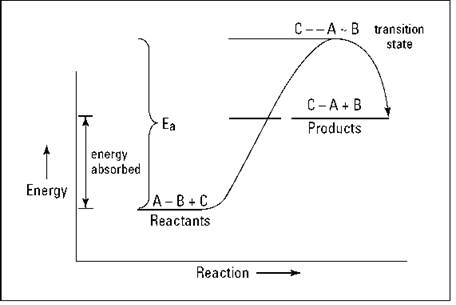
Oil forms a barrier between the pan and the egg, and also conducts heat better, what actually keeps an egg from sticking is the steam produced by the egg cooking.
An endothermic reaction takes in energy from the surroundings;
Exothermic reactions transfer energy to the surroundings, and this energy is usually heat energy, they cause the surroundings to heat up.
Is frying eggs without using oil or butter even still considered frying?
Then, cover the pan to allow the eggs to cook with the steam generated from the heat, ensuring that they are perfectly cooked without the need for a drop of oil.
Endothermic absorbs heat, and exothermic produces heat.
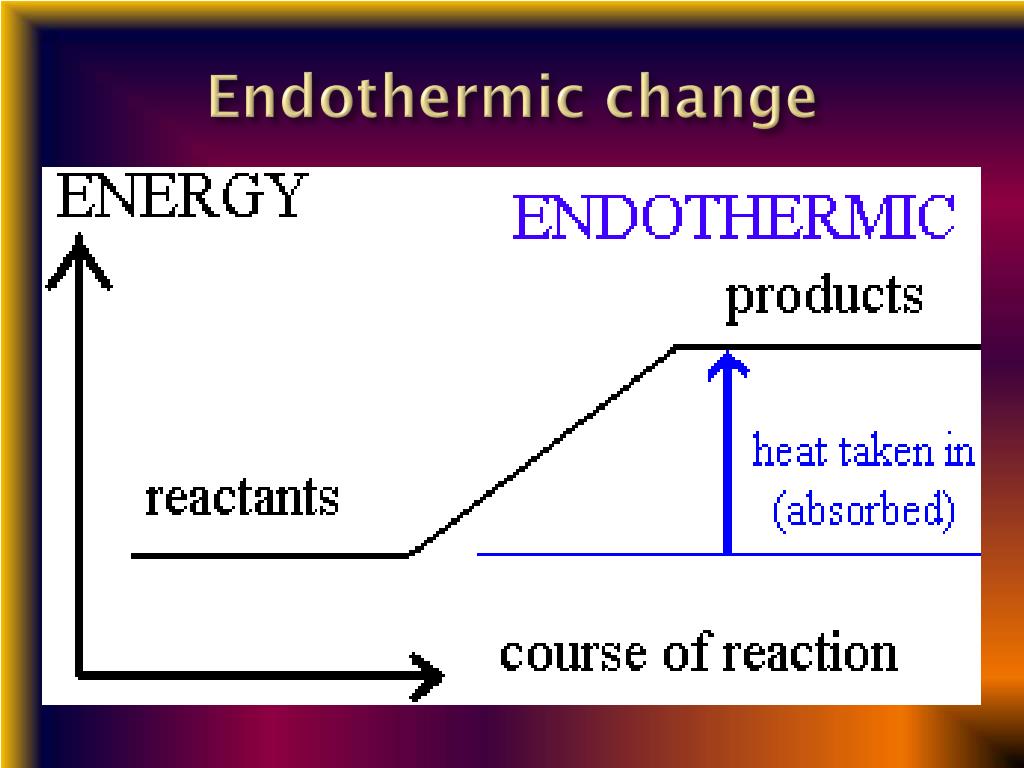
There must be heat added or absorbed from in summary, endothermic consists of energy or heat being absorbed from its surroundings, and exothermic involves energy or heat being released.
Baking bread and cooking an egg are examples of.?
During an endothermic reaction in a beaker if we are part of the surroundings and touched the beaker, it would feel _.
Are there any general trends the energy stored in the molecules(specifically in the bonds) is lower before the reaction in an endothermic reaction and higher in an exothermic reaction.
Who could resist a crispy, crunchy cutlet with a tender juicy center?
It's less messy than deep frying, too—get the basic information right here and start frying tonight.

This article explains the differences between the two reaction types.
Every change that you see in a natural system involves the transaction of energy.
Thermodynamics is the branch of physics devoted to studying these changes.

Photosynthesis and endothermic reactions sweat the process of cooking is a endothermic reaction as heat is absorbed while cooking the food.
Photosynthesis is an example of an endothermic chemical reaction.
Photosynthesis is an example of an endothermic chemical reaction. Is Cooking An Egg In A Frying Pan Exothermic Or Endothermic. In this process, plants use the energy from the sun to convert carbon.Resep Ayam Kecap Ala CeritaKuliner5 Cara Tepat Simpan TelurResep Beef Teriyaki Ala CeritaKulinerTernyata Asal Mula Soto Bukan Menggunakan DagingJangan Sepelekan Terong Lalap, Ternyata Ini ManfaatnyaAmpas Kopi Jangan Buang! Ini ManfaatnyaSejarah Prasmanan Alias All You Can EatBlack Ivory Coffee, Kopi Kotoran Gajah Pesaing Kopi Luwak5 Kuliner Nasi Khas Indonesia Yang Enak Di LidahBakwan Jamur Tiram Gurih Dan Nikmat
Komentar
Posting Komentar