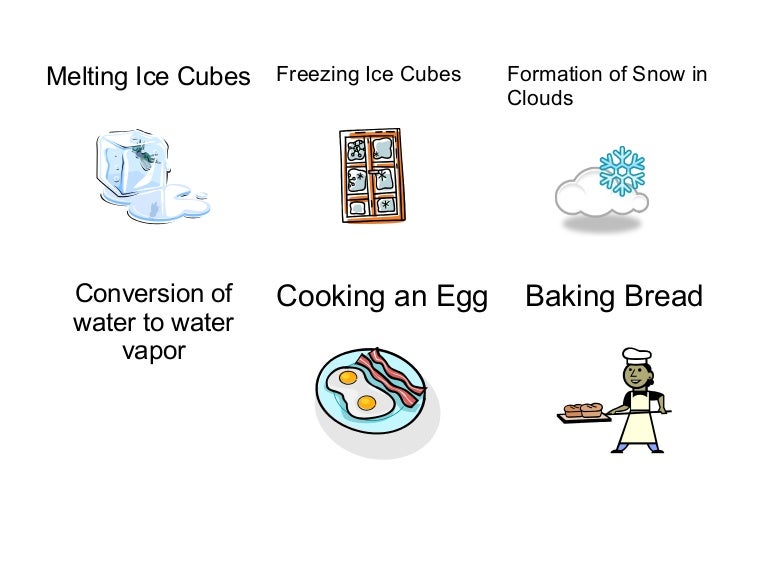
Is Cooking An Egg Exothermic Or Endothermic The Answer Is Quite Similar In This Case As Well.
Is Cooking An Egg Exothermic Or Endothermic. The Reaction Absorbs The Heat Energy That Is Being Produced By The Stove.
SELAMAT MEMBACA!
Cooking of an egg is an endothermic reaction.
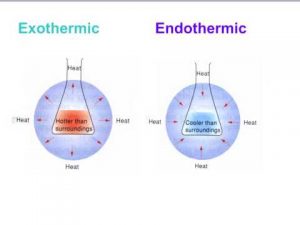
When considering whether a reaction is endothermic or exothermic, it is vital to separate the reaction system from the environment.
Cooking an egg is an endothermic process because the heat from the stove is inputted to the egg so it can cook.
Endothermic, heat absorbed or in something, exothermic, heat being released or sent out.

There must be heat added or absorbed from the environment to cook the egg or any other food item.
An egg cooking is an endothermic reaction.
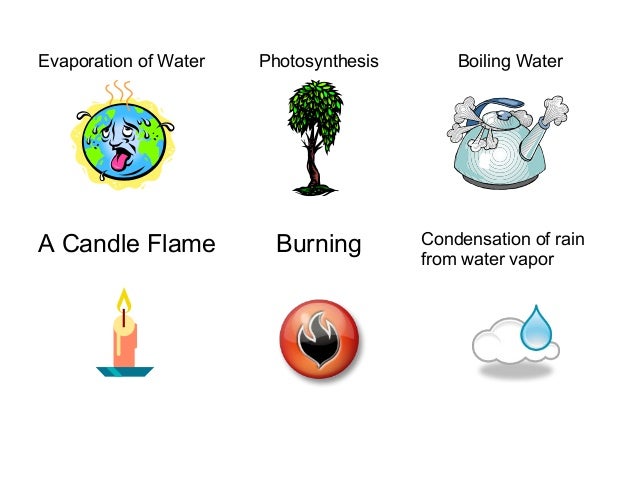
A candle burning and plaster & water combining and becoming warm are exothermic reaction.

It is a type of.
When you cook there is heat involved which enforces reactions between different materials.
Additionally it is essentially the most durable and gives the best cooking results.

The difference between the words themselves and the prefixes give another hint as to their endothermic must be supplied with heat and is basically the opposite of exothermic.
An everyday reaction is in the cooking of an egg.
Exothermic vs endothermic processes in physics.

Energy is absorbed from the pan to cook the egg.
An exothermic process is a procedure where energy is produced as an output of the process when it's done.
Usually the energy forms as heat energy.
The answer is quite similar in this case as well.
The egg absorbs heat from the.
Heat leaves the system, heat is a product.
/FriedEgg-58e5117d5f9b58ef7e550238.jpg)
Endothermic and exothermic reactions refer to the absorption or release of heat.
In any given reaction, heat is both absorbed and released.
Cooking an egg is an endothermic process because added energy makes it cooked.

In this reaction, energy is absorbed.
Is exothermic hot or cold?
An exothermic process releases heat, causing the temperature of the immediate surroundings to rise.

An everyday reaction is in the cooking of an egg.
There must be heat added or absorbed from in summary, endothermic consists of energy or heat being absorbed from its surroundings, and exothermic involves energy or heat being released.
An exothermic reaction occurs when the temperature of a system increases due to the evolution of heat.

Outside) describes a process or reaction that releases energy from the system to its surroundings, usually in the form of heat, but also in a form of light (e.g.
A spark, flame, or flash), electricity (e.g.
This topic is for explaining the difference between endothermic and exothermic reactions.

Rusting iron, settling, chemical bonds, explosions, nuclear fission are a few examples.
An endothermic process is any process or reaction which absorbs or assimilates power or energy from its environment, for the most part as heat.
The inverse of an endothermic process is an exothermic process, one that discharges, gives out energy or power as warmth.
It all has to do with the direction of heat movement, or heat transfer.
If heat transfers from the surroundings into a system, it is an endothermic process.
Compare and contrast endothermic and exothermic reactions, and learn how heat is either absorbed or released during chemical reactions.
The reaction absorbs the heat energy that is being produced by the stove.
Get the definition of an endothermic reaction and endothermic reaction examples.
Learn how the terms endothermic and endergonic are related.
Because heat is absorbed, endothermic reactions feel.
Baking bread and cooking an egg are examples of.?
During an endothermic reaction in a beaker if we are part of the surroundings and touched the beaker, it what instrument would you use to detect an endothermic or exothermic reaction?
This article explains the differences between the two reaction types.
Everything that happens in nature is some form of an energy change and depending on the direction of energy flow, that change may be classified as an.
Energy is absorbed from the pan to cook the egg.

An exothermic reaction is a process that releases energy to the surroundings, usually in the form of heat.
Can we determine whether a reaction is endothermic or exothermic?
For example if we are given the following reactions can we determine weather these reactions are endothermic or exothermic or do we have to just memorize this?
Let's break these words down.
Starting with endothermic, the prefix endo means within, inner when deciding whether a reaction is endothermic or exothermic, ask yourself if more heat is added or taken away in a particular reaction.
Enthalpy is the amount of energy in a system, and according to the 1st law of thermodynamics, energy cannot be created or destroyed, only an endothermic reaction is a reaction where the system absorbs energy in the form of heat, making the surroundings feel colder.

This explains why the closer you get the equator, or.
Efek Samping Mengkonsumsi Bawang Merah Yang Sangat Berbahaya Bagi TubuhAsi Lancar Berkat Pepaya Muda5 Rahasia Tetap Fit Saat Puasa Ala KiatSehatkuTernyata Madu Atasi InsomniaAwas!! Ini Bahaya Pewarna Kimia Pada MakananTernyata Rebahan Mempercepat Penuaan7 Makanan Sebabkan Sembelit5 Khasiat Buah Tin, Sudah Teruji Klinis!!6 Khasiat Cengkih, Yang Terakhir Bikin HebohJam Piket Organ Tubuh (Limpa)Internal heating, if you will. Is Cooking An Egg Exothermic Or Endothermic. This explains why the closer you get the equator, or.
Cooking of an egg is an endothermic reaction.

If the system cools down, that means heat is being released, and the reaction taking place is an exothermic reaction.
In this case of cooking an egg.
Before i start my answer, i should note that (as mentioned by another person who answered), the process you are describing (or rather, that i presume that you are describing) is more of a question of thermodynamics than chemistry.
Cooking an egg is an endothermic process because added energy makes it cooked.
An egg without heats stays an (uncooked) egg.
Which answer defines exothermic reaction?

An egg cooking is an endothermic reaction.
A candle burning and plaster & water combining and becoming warm are exothermic reaction.
Salt being added to water, with no change in temperature neither endothermic nor exothermic reaction.

This is your cooked egg (and why it is generally not reversible).
An everyday reaction is in the cooking of an egg.
An endothermic reaction will cool their surroundings because the reactions draw the heat energy into themselves.

An endothermic reaction occurs when energy is absorbed from the surroundings in the form of heat.
Conversely, an exothermic reaction is one in which energy is released from the system into the surroundings.
The terms are commonly used.
An everyday reaction is in the cooking of an egg.
There must be heat added or absorbed from in summary, endothermic consists of energy or heat being absorbed from its surroundings, and exothermic involves energy or heat being released.
Endothermic and exothermic reactions refer to the absorption or release of heat.
![Why is Cooking an Endothermic Process? [A Quick Explanation]](https://ml2kdkdjqd5j.i.optimole.com/NSdRypY-wmf2vtDN/w:auto/h:auto/q:100/https://dwellhack.com/wp-content/uploads/2020/06/Why-is-Cooking-an-Endothermic-Process-Dwell-Hack.jpg)
Examples include light and sound.
In general, reactions involving energy may be classified as endergonic or exergonic, an.
This is the currently selected item.
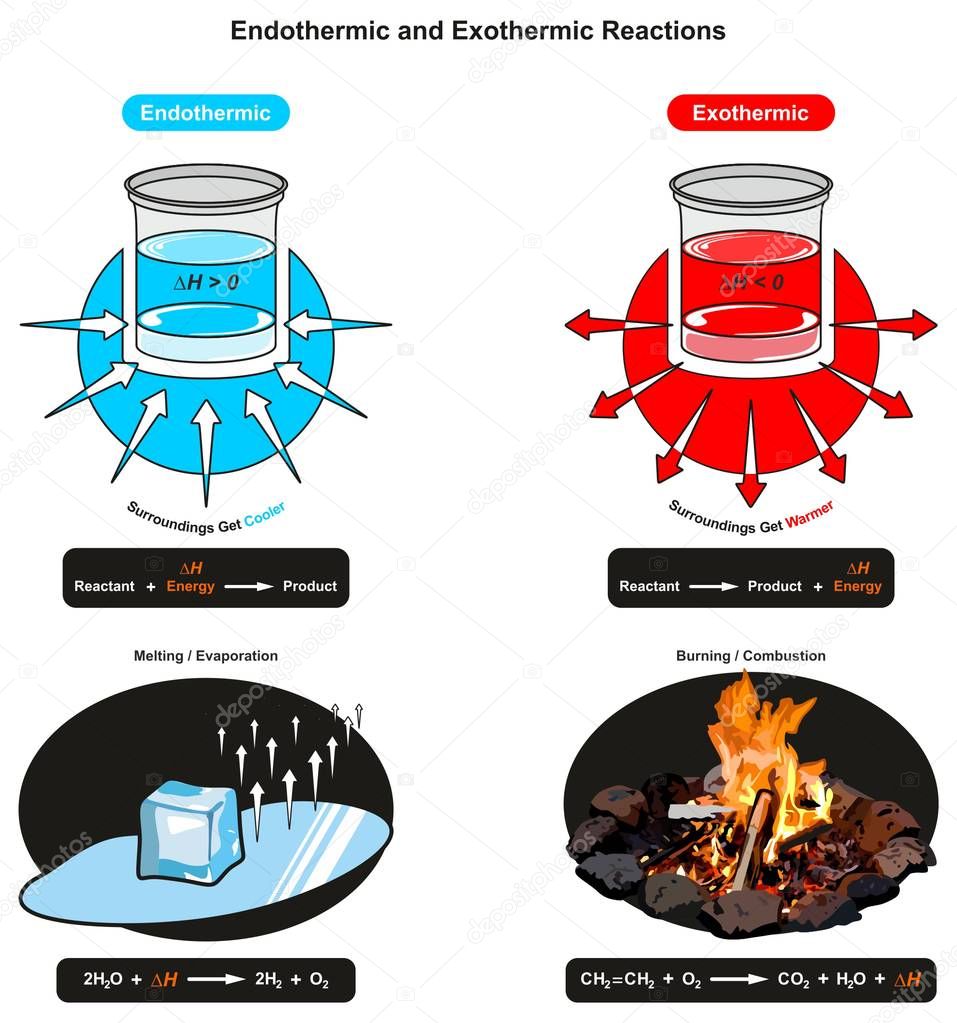
Freezing water into ice cubes.
(when the cake is in the over the oven must keep putting more heat in to cook the.
The exothermic reaction produces energy so that the surrounding feels hot.

The answer is quite similar in this case as well.
The egg absorbs heat from the.
These reactions are the opposite of an endothermic reaction.

Faqs on exothermic and endothermic reactions:
What will happen with the rate of an exothermic.
This chemistry video tutorial focuses on endothermic and exothermic reactions.
An endothermic reaction takes in energy from the surroundings;
Exothermic reactions transfer energy to the surroundings, and this energy is usually heat energy, they cause the surroundings to heat up.
Just like a bonfire keeping everyone warm.
The opposite of an endothermic reaction is an exothermic reaction.
Endothermic and exothermic reactions are fundamentally different.
This article explains the differences between the two reaction types.

Any chemical reaction or change in which energy in the form of heat is released.
On a molecular level, chemical as a result, the temperature of the ice rises and it turns into water!
Basically, melting ice is an endothermic reaction because the ice absorbs (heat).

Energy is absorbed from the pan to cook the egg.
The word exothermic is formed by exo.
An exothermic reaction occurs when the temperature of a system increases due to the evolution of heat.
A system of reactants that absorbs heat from the surroundings in an endothermic reaction has a.
Baking bread and cooking an egg are examples of.?
A chemist is doing an endothermic reaction.

Can we determine weather these reactions are endothermic or exothermic or do we have to just memorize this?
In short you have to know something.
The energy stored in the molecules(specifically in the bonds) is lower before the reaction in an endothermic reaction and higher in an exothermic.

When an egg is cooked over a stove the reaction taking place is endothermic.
The reaction absorbs the heat energy that is being produced by the stove.
An endothermic process is any process or reaction which absorbs or assimilates power or energy from its environment, for the most part as heat.
In the investigation on exothermic and endothermic reactions learners work with concentrated sulfuric acid.
They must work in a well ventilated observe whether the reaction is endothermic or exothermic, either by feeling whether the side of the test tube gets hot or cold, or using a thermometer.
Learn about exothermic and endothermic reactions and the transfer of energy with gcse bitesize combined science (aqa).
In other words, exothermic reactions release heat as a product, and endothermic reactions consume heat as a reactant.
So reaction enthalpy changes (or reaction heats) are a useful way to measure or predict chemical change.
But they're just as useful in dealing with physical changes, like.
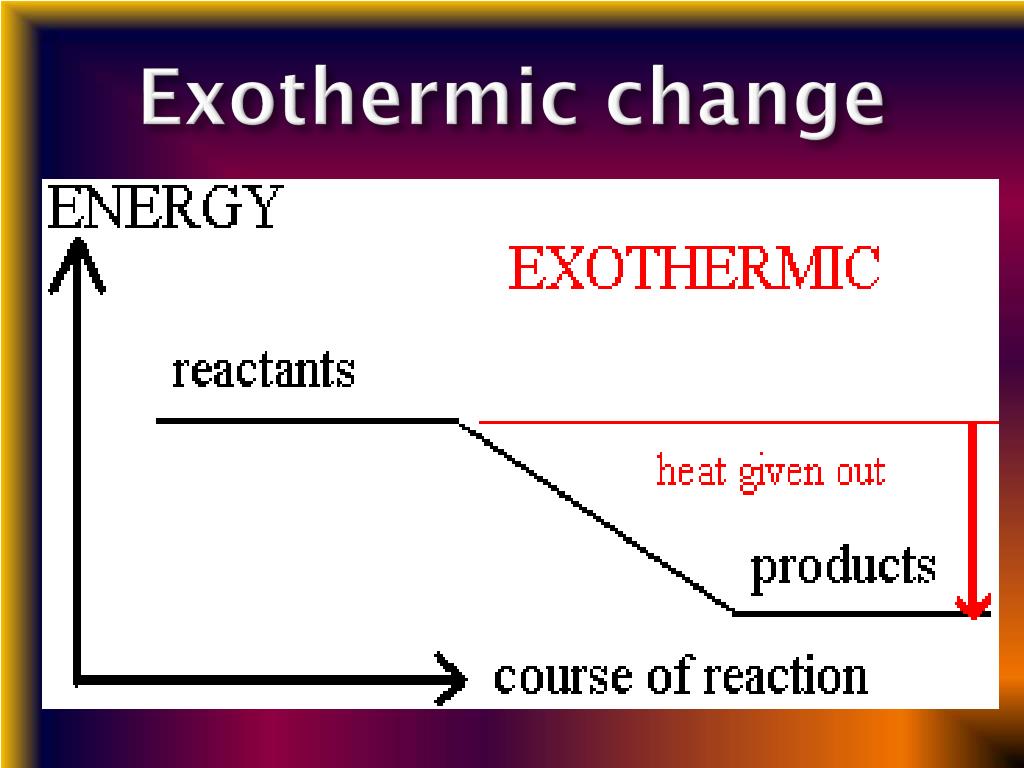
Learn how endothermic reactions are different from exothermic reactions.
You might not have realized cooking an egg was an endothermic reaction.
However, energy from the pan is absorbed to cook the egg in this endothermic reaction.

Therefore, the change in enthalpy is positive, and whether a reaction is endothermic or exothermic depends on the direction that it is going;
Some reactions are reversible, and when you.
Therefore, the change in enthalpy is positive, and whether a reaction is endothermic or exothermic depends on the direction that it is going; Is Cooking An Egg Exothermic Or Endothermic. Some reactions are reversible, and when you.Foto Di Rumah Makan PadangTips Memilih Beras BerkualitasAyam Goreng Kalasan Favorit Bung KarnoPlesir Ke Madura, Sedot Kelezatan Kaldu Kokot MaduraWaspada, Ini 5 Beda Daging Babi Dan Sapi!!Jangan Ngaku Penggemar Burger Kalau Tak Tahu Sejarah Ditemukannya Hamburger5 Cara Tepat Simpan TelurResep Pancake Homemade Sangat Mudah Dan EkonomisTernyata Makanan Ini Sangat Berbahaya Kalau Di PenjaraTernyata Terang Bulan Berasal Dari Babel
Komentar
Posting Komentar